An ultrasound imaging technique called passive cavitation imaging was able to create an image and estimate the amount of a drug that crossed the blood-brain barrier to reach a specific location in the brain, according to a study by NIBIB-funded University of Washington bioengineers. The technique monitors the activity of microbubbles, microscopic bubbles that help create clearer ultrasound images using detectors to estimate the effects they have on different biological structures, in this case, the brain.
The brain is responsible for a person’s most vital functions; Therefore, the brain is protected by a difficult-to-penetrate boundary called the blood-brain barrier (BBB). However, there are times when disease treatments must go beyond the BBB to treat a disease that has invaded the brain.
“Researchers are developing non-invasive approaches that use microbubbles and focused ultrasound waves to briefly disrupt the blood-brain barrier (FUS-BBBD),” explains Randy King, Ph.D., director of the ultrasound program at the National Institute of Biomedical Imaging and Bioengineering (NIBIB). “As new techniques are established to permeate the BBB, we need complementary methods to monitor the delivery of treatments and ensure they are delivered safely to the site of brain disease.”
Hong Chen, Ph.D., assistant professor of biomedical engineering and radiation oncology at Washington University in St. Louis, explained that FUS-BBBD is achieved by concentrating, or focusing, sound waves emitted from a device, called a transducer, at a specific location. Chen compared the concept to focused sunlight passing through a magnifying glass to a localized point. In FUS, ultrasound waves are focused with specially designed ultrasound generators and help focus the opening of the BBB to a specific location in the brain.
Microbubbles can be injected into the bloodstream to amplify the effects of ultrasonic waves on blood vessels, thereby opening the BBB in a localized location. Researchers can store drugs for treatments inside the microbubbles. The pressure generated by ultrasound pulses can cause microbubbles in the bloodstream to expand, contract, and eventually burst. When microbubbles rupture in the blood vessels of the BBB, the pressure created gently massages the blood vessels and makes them permeable to medications injected into the bloodstream or carried by the microbubbles.
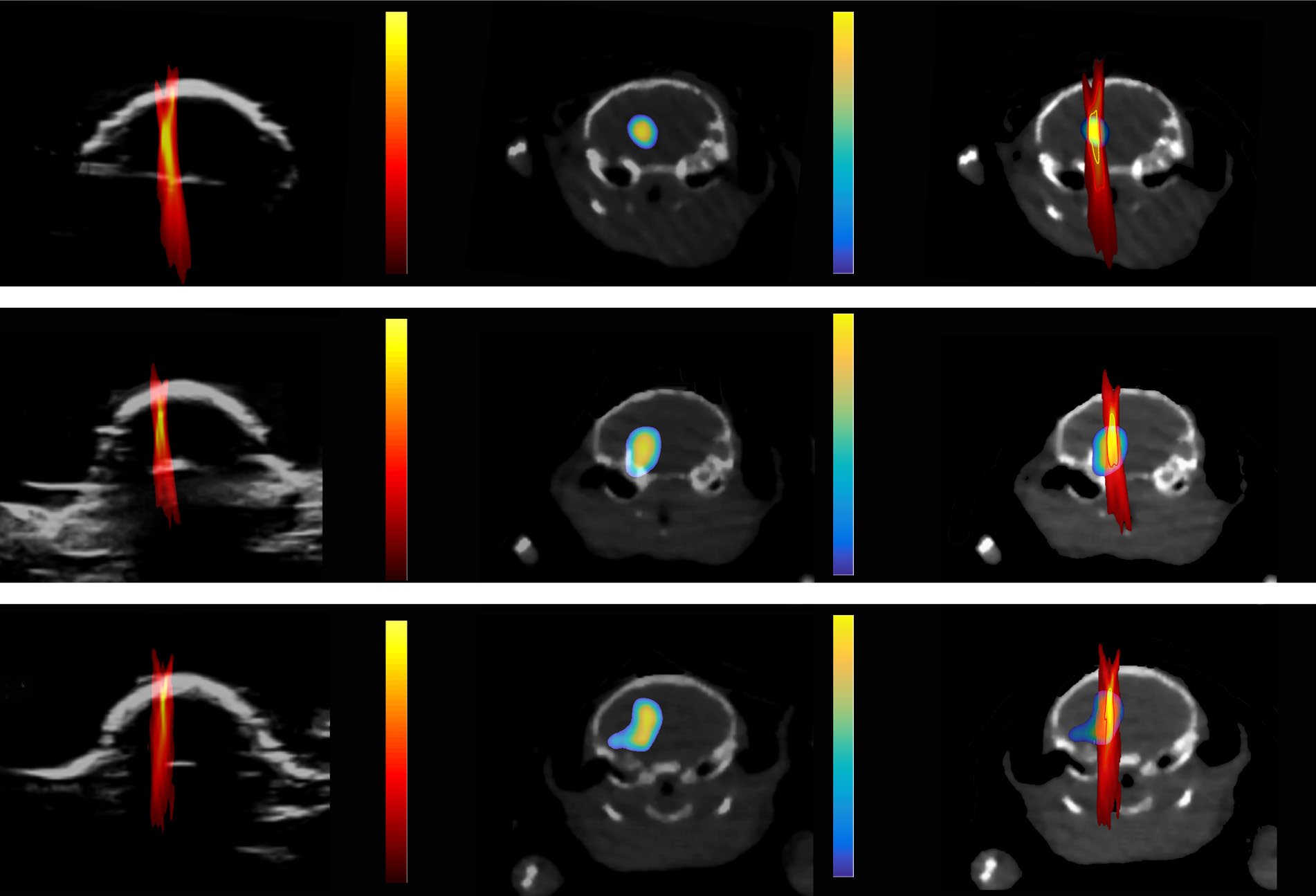
To track the amount and location of the drug delivered by FUS-BBBD, the scientists labeled nanoparticles with a radioactive tracer and injected them into the blood circulation after FUS treatment. Positron emission tomography (PET) and computed tomography (CT) imaging were then used to measure and visualize the precise number and location of the nanoparticles in the brain, respectively.
“The disadvantages of using PET/CT imaging are the exposure associated with radioactivity and higher costs,” Chen explained. The team wanted to find a cheaper and safer way to control where medications go after crossing the BBB, so they explored a technique called cavitation dose painting using passive cavitation imaging (PCI). The study was published in Scientific Reports.
Yaoheng Yang, lead author of the study and a graduate student at the University of Washington, explained that PCI monitors the behavior of microbubbles in the ultrasound field and does not rely on a radioactive particle like PET/CT images. Recording the dynamic behavior of the small bubbles allows researchers to create a detailed image that can track the location and amount of drug during FUS-BBBD treatment. But the question remained whether PCI images would correlate with PET/CT images.
“Our results demonstrated a pixel-by-pixel correlation between PET and PCI images,” Chen said. Chen is hopeful that this advancement in imaging will push the field of drug delivery toward better precision medicine. The team calls the new technique cavitation dose painting because it is like “painting by numbers” for drug delivery.
With the new $1.6 million NIBIB grant, Chen plans to fully integrate PCI with FUS-BBBD, so that the physician has full control over medication administration. “With our new technique, we can predict exactly where a drug will go and how much of it will be released when it gets there. It helps us minimize the damage a drug does to healthy parts of the brain and avoid ineffective treatments when it misses its intended target,” Chen said.
This work was supported in part by the Children’s Discovery Institute at Washington University and St. Louis Children’s Hospital (grant number MC-II-2017-661), the American Cancer Society (grant number IRG-15-170-58), and National Institutes of Health (NIH) grant R01MH116981 and R01EB027223.
Yaoheng Yang and others, Cavitation Dose Painting for Focused Ultrasound Induced Blood Brain Barrier DisruptionScientific Reports (2019).