Macrophages that normally support tumor growth express a unique surface protein that enables targeted cancer therapy
Macrophages are white blood cells that accumulate in tumors, where they help cancer progress. Now scientists have identified a surface protein found only on macrophages that reside in tumors, exposing a target for precise tumor treatments.
Most tumors contain macrophages, a type of immune cell, which help tumor growth through several mechanisms. Tumor-associated macrophages (TAMs) act to stimulate the growth of new blood vessels, remodel the extracellular matrix to promote metastasis, and enhance drug resistance.
Despite the role of TAMs in aiding tumor growth, due to their high concentration in tumors, researchers have sought ways to exploit these cells to target tumor therapies. However, a major barrier to achieving this goal is how to distinguish TAMs from macrophages in normal organs. Now, NIBIB-funded Hongbo Pang, Ph.D., an assistant professor at the University of Minnesota School of Pharmacy, and his collaborators have identified a protein found only on the surface of TAMs. The work, which aims to guide specific targeting of tumor therapies, is reported in the May issue of the Journal of Controlled Release. 1.
“Delivering drugs to tumors, while avoiding exposure to healthy tissues, is a central goal in cancer treatment,” says David Rampulla, Ph.D., NIBIB program director in Drug and Biologics Delivery Systems and Devices. “Identifying a cell surface protein specific only to macrophages in tumors and not healthy tissues is an exciting finding with the real possibility of improving the specificity and potency of therapies for a wide range of cancers.”
Pang and his colleagues previously used a technique called phage display to identify a marker found only on the surface of TAMs. Phage display involves placing thousands of random DNA fragments into phages, which are viruses that infect and grow in bacteria. The piece of DNA from each phage produces a peptide, a small piece of protein, which is displayed on the surface of the phage. The result is an extensive library of random peptides on the surface of phages. The phage library was then exposed to a macrophage cell line, leading to the identification of a macrophage-binding peptide called CRV.
In this study, Dr. Pang and his team injected CRV into mice carrying a variety of tumor types. CRV successfully targeted and bound to TAMs in tumor tissue while avoiding surrounding healthy tissues. To bind to TAMs, CRV had to move from the tumor’s blood vessels into the tumor tissue itself, a process called extravasation. The rapid extravasation of CRV to reach TAMs was an extremely encouraging sign that this peptide has the potential to deliver therapeutic cargo to solid tumors.
The team validated that CRV binds to a receptor on the surface of TAMs called RXRB. Additional analyzes confirmed that RXRB is not found on the surface of macrophages in normal organs and is therefore a distinct marker found only in TAMs.
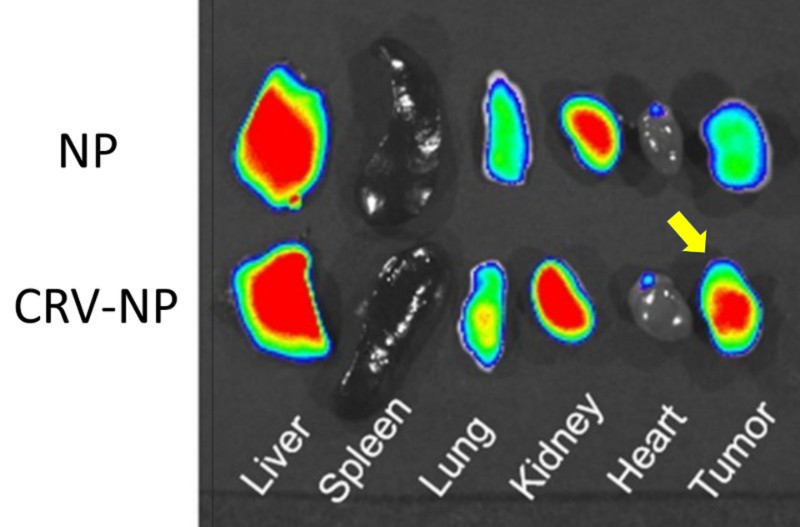
To test the idea of using the system to deliver anti-tumor drugs, the team attached CRV to a nanoparticle, which could potentially deliver chemotherapy drugs to the TAMs. The CRV nanoparticle was injected into mice bearing mouse mammary tumors. CRV improved delivery of nanoparticles to solid tumors.
“The results demonstrate that we have defined a potentially novel target in TAMs to improve TAM-based cancer therapy,” says Pang. “This opens up the possibility of a number of therapies targeting TAMs, ranging from the highly specific delivery of chemotherapy to tumors, to the development of TAM-binding molecules that could potentially reverse TAMs from being tumor promoters to potent antitumor weapons.”
This work was supported by grants from the National Cancer Institute (R01CA214550 and R01CA188883) and the National Institutes of Biomedical Imaging and Bioengineering (R21EB022652). Dr. Pang is also affiliated with Sanford Burnham Prebys Medical Discovery Institute, La Jolla, CA.