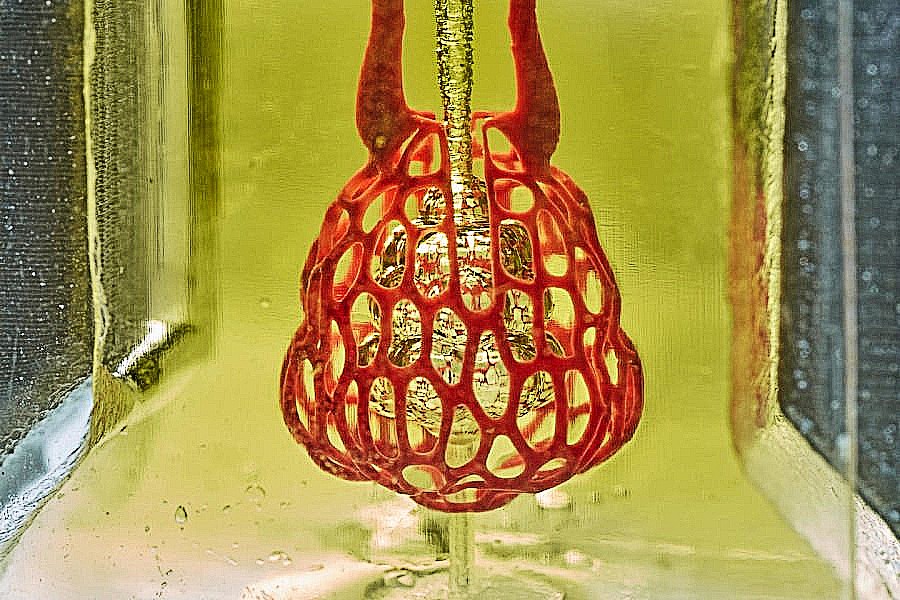
Bioengineers have developed a 3D printing technique that creates interactive networks for transporting air, blood and other body fluids, an important step toward 3D printed organ replacement.
In the field of tissue engineering, a major obstacle to creating durable functional tissues has been the difficulty of printing the complex networks of blood vessels, airways, and other systems that can reliably supply the tissue with necessary nutrients and remove waste products.
Now, bioengineers led by Jordan Miller, Ph.D., assistant professor of bioengineering at Rice University, and Kelly R. Stevens, Ph.D., assistant professor of bioengineering and pathology at the University of Washington, have developed a novel 3D printing technique that represents a significant advance in the push to provide replacement organs. The team included 15 collaborators from Rice, UW, Duke University, Rowan University and Nervous System, a company in Somerville, Massachusetts. The results appear in the May issue of Science.1
“Not surprisingly, it took a large interdisciplinary team including bioengineers, tissue engineers, pathologists and computational scientists to devise the unique approach needed to build the intricate system needed to deliver nutrients to bioprinted tissues,” said Seila Selimovic, director of the NIBIB Program in Tissue Engineering, which partially funded the study. “It will be exciting to see how this revolutionary advance develops in terms of our ability to bioprint and transplant working organs.”

The approach added a unique level of precision and detail to 3D bioprinting that had not been possible before. The group used a technique called stereolithographic printing, which is primarily used with resins that harden into the desired pattern when hit with a laser. However, the hard resins could not support living cells. By adding laser-sensitive food dyes to the biogel material needed to support living cells, the team was able to extend to biogels the levels of complexity and printing scale previously achieved with resins alone. The added dyes were the key to depositing and solidifying many consecutive thin layers of biogel with the intricate vascular patterns of an organ.
The team printed a series of organ-like structures, including the microstructure of the lung, where vessels carrying air and blood wrap around each other. This is the interaction in the lungs that allows the air we breathe to be transferred to the bloodstream and distributed throughout the body. In tests under the pressures found in the human lung, the lung-mimicking structure was able to expand with the influx of oxygen and the network-like structure of blood vessels surrounding the “breathable” air sac was able to absorb the oxygen.
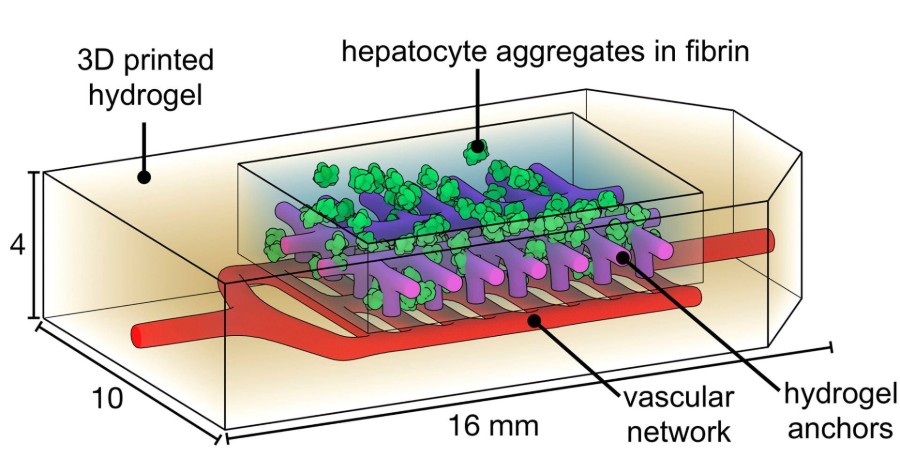
The team also printed liver-like structures, which survived when implanted into mice with liver injury, as well as blood vessels with functional valves that mimic the valves that control the movement of blood and other fluids in the heart, leg veins and lymphatic system.
“The design of this system is an important step towards tissue engineering of organs such as the liver, which is an extremely complex organ with many functions,” explains Dr. Stevens. “The ultimate goal would be to one day be able to provide bioprinted livers to thousands of people on transplant waiting lists.”
Although this is an ambitious goal, the team believes that the progress they continue to make could make organ bioprinting an important component of medicine within the next twenty years.
The work was supported by the Robert J. Kleberg, Jr. and Helen C. Kleberg Foundation, the John H. Tietze Foundation, the National Science Foundation (1728239, 1450681, and 1250104), the National Institutes of Health (F31HL134295, DP2HL137188, T32EB001650, T32GM095421, and DP5OD019876) and the Gulf Coast Consortia.